|
9/19/2023 0 Comments F atomic radius 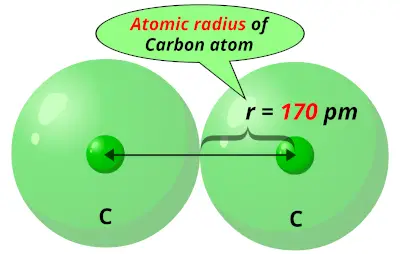
Smallest and Largest Atomic Radiusįrancium has the largest atomic size on the periodic table, and helium has the smallest atomic size. The Trend on a GraphĪs shown in the graph below, the atomic radius is largest at the first element in each period, and it decreases down each period. As electron cloud sizes increase, so do atomic radii. This is because between each group, electrons occupy successively higher energy levels. Group Trendĭown a group, atomic radii increase. This is why the difference in atomic radii decreases down each period. F and are smaller and they have less shells. One thing to note is that the effect of the attraction between the positively charged nucleus and the electrons is slightly countered by the repulsion of electrons as they are successively added. Thus, the radius of fluoride ions is larger than that of fluorine atoms. This increased positive charge attracts or pulls, the electrons in closer to the nucleus, decreasing the atomic radius. It is located in group 17 and period 2 in the modern periodic table. Theamount of energy requiredto remove the outermost electronfrom a gaseousatom in the ground state isknown as (A)firstionization energy (B) activation energy (C) conductivity (D)electronegativity. Fluorine is the 9 element on the periodic table. Whichnoble gas has the highest first ionization energy (A) radon (B)krypton (C) neon (D)helium (B) increases. What is the position of Fluorine in the Periodic Table Fluorine is a chemical element with the symbol F and atomic number 9. Down the period, however, the number of protons also increases. Fluorine is a chemical element with symbol F and atomic number 9. This is because while the number of electrons increases down the period, they only add to the same main energy level, and therefore do not expand the electron cloud. All values of radii are given in picometres (pm). Follow the appropriate hyperlinks for literature references and definitions of each type of radius. For example, ionization energy, electronegativity, and of course atomic radius which we will discuss now. There are several other ways ways to define radius for atoms and ions. There are many trends on the periodic table. Let’s break down the trend into its period and group trends. Liquid fluorine is soluble in liquid oxygen and ozone. Atoms decrease in size across the period and increase in size down the group. Atomic mass of fluorine is 18.998 u, which makes it the lightest halogen on the periodic table. 
Atomic Radius Trend on the Periodic TableĪtomic radii increase toward the bottom left corner of the periodic table, with Francium having the largest atomic radius. Thus the atomic radius is measured as shown in the diagram below. This is because the borders of orbitals are quite fuzzy, and they also change under different conditions. While your initial thought may have been to measure the distance from the center of an atom’s nucleus to the edge of its electron cloud, this is inaccurate and not feasible. The atomic radius is measured as half the distance between two nuclei of the same atoms that are bonded together. Let’s discuss the definition of the atomic radius, also called atomic size, and the atomic radius trend on the periodic table. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
